|
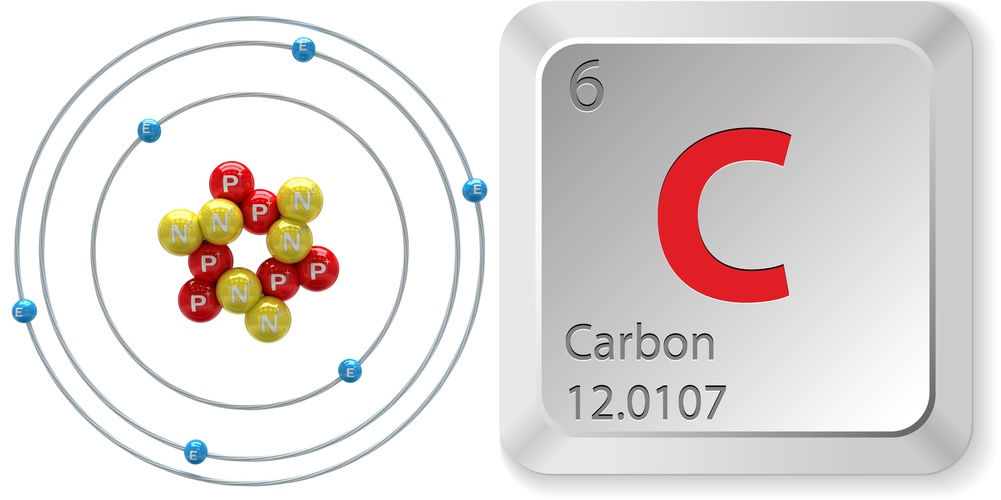
Atomic Number (number of protons in the nucleus): 6 Atomic Weight (average mass of the atom): 12.0107 Density: 2.2670 grams per cubic centimeter Number of isotopes: 15 total; two stable isotopes, which are atoms of the same element with a different number of neutrons Most common isotopes: carbon-12 (6 protons, 6 neutrons and 6 electrons) and carbon-13 (6 protons, 7 neutrons and 6 electrons) Melting Point: 6,422 degrees Fahrenheit (3,550 degrees C) Boiling Point: 6,872 F (3,800 C) (sublimation)
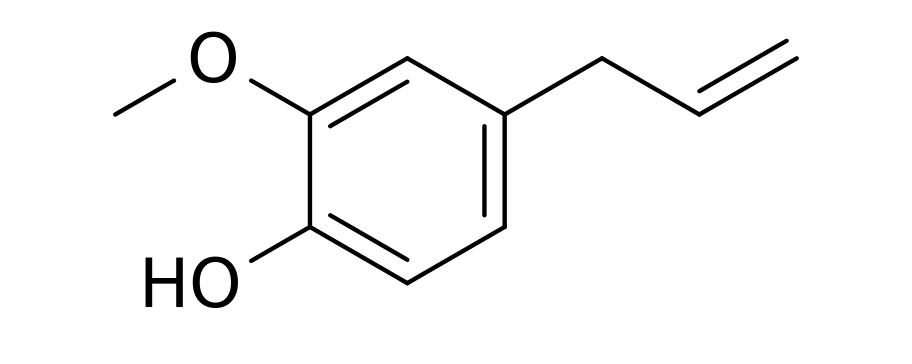
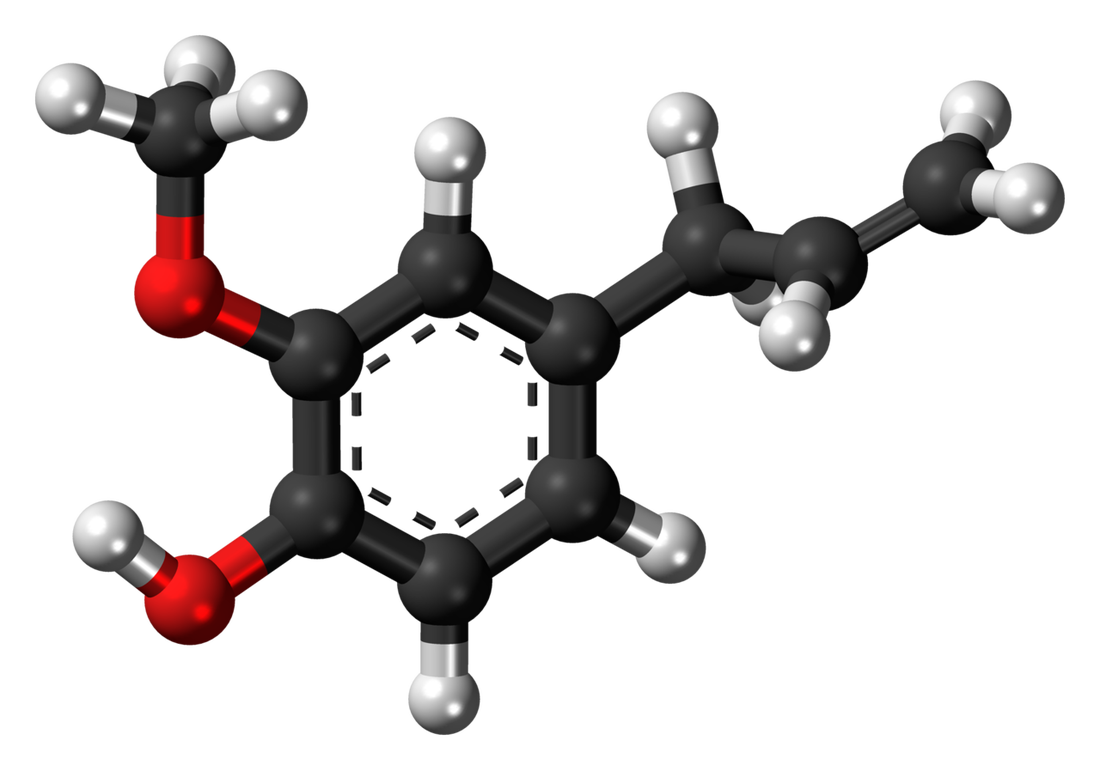
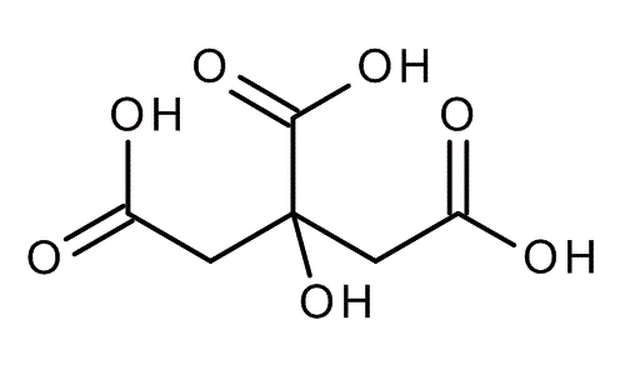
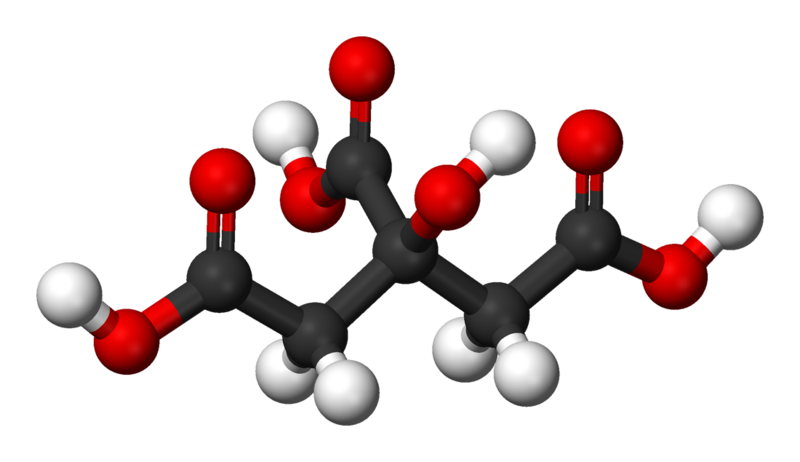
C10H12O2Eugenol is a phenylpropene, an allyl chain-substituted guaiacol. Eugenol is a member of the phenylpropanoids class of chemical compounds. It is a colourless to pale yellow oily liquid extracted from certain essential oils especially from clove oil, nutmeg, cinnamon, basil and bay leaf. It is present in concentrations of 80–90% in clove bud oil and at 82–88% in clove leaf oil. Uses of Eugenol:Eugenol is used in perfumes, flavorings, and essential oils. It is also as a local antiseptic and anaesthetic. Eugenol can be combined with zinc oxide to form a material – known as zinc oxide eugenol – which has restorative and prosthodontic applications in dentistry. For example, zinc oxide eugenol is used for root canal sealing. Attempts have been made to develop eugenol derivatives as intravenous anesthetics, as an alternative to propanidid which produces unacceptable side effects around the site of injection in many patients. It can be used to reduce the presence of Listeria monocytogenes and Lactobacillus sakei in food. It is also used in manufacturing stabilizers and antioxidants for plastics and rubbers. It is one of many compounds that is attractive to males of various species of orchid bees, which apparently gather the chemical to synthesize pheromones; it is commonly used as bait to attract and collect these bees for study. It also attracts female cucumber beetle. It is recently discovered that eugenol and isoeugenol, floral volatile scent compounds, are catalyzed by single type of enzyme in Gymnadenia species and gene encoding for this enzyme is first functionally characterized gene in this species so far. Clove oil is growing in popularity as an anaesthetic for use on aquarium fish as well as on wild fish when sampled for research and management purposes. Where readily available, it presents a humane method to euthanise sick and diseased fish either by direct over-dose or to induce sleep before an overdose of ethanol. Eugenol Canada. Eugenol USA. Eugenol Mexico. Eugenol Latin America. Eugenol Africa. Eugenol India. Eugenol China. Eugenol Australia. Eugenol Europe. Eugenol Middle East. Eugenol Japan. Eugenol Korea. Eugenol France. Eugenol Spain. Eugenol Italy. Eugenol Germany.Citric Acid Latin America. Acido Citrico Latinoamerica. Acid Citrique Amerique Latine. Citric Acid International. Citric Acid Europe. Citric Acid Africa. Citric Acid Australia. Citric Acid Asia. Citric Acid India. Citric Acid Middle East. Acido Citrico Centroamerica. Citric Acid Caribbean.Chemical Formula CITRIC ACID occurs widely in nature and derives its name from the citrus fruits where its presence imparts a refreshing tangy taste. It also occurs in many other fruits, natural products such as milk and in various plant products. Typical levels of CITRIC ACID found in some common fruits are shown below: Citric Acid Production ProcessManufactured by the process of fermentation of a carbohydrate substrate from saccharose or dextrose. It was not until the 1920's that researchers in the US and Europe were able to perfect the large-scale, commercial production of citric acid by the microorganism Aspergillus niger, a mould belonging to the same family as the penicillins. The main raw materials used in citric acid production are carbohydrates such as sucrose and molasses. Production involves surface and deep fermentation techniques, in many ways similar to those used for beer production... Pure white crystalline citric acid is produced from the fermentation broth in two forms, anhydrous and monohydrate. Recovery techniques used may include precipitation, evaporation and crystallisation. Salts of citric acid are produced by neutralisation with the appropriate alkali. The salt most widely used is sodium citrate. Applications: Foods & Beverages Canned Foods & Vegetables: Allows reduction of retorting temperature. Preserves flavor, appearance and consistency. Dairy Products & Ice Creams: Provides acidity and enhances flavors. As emulsifying agent in ice creams to keep fats from separating.Fats & Oils: Provides acidity, enhances flavors and acts as preservative and antioxidant. Jams & Jellies: pH adjustment for optimal pectin jelling. Soft Drinks: Imparts fine tangy flavor. Sequesters harmful heavy metal ions. Wine: Prevents or dissolves ferric turbidity caused by tannin-iron or phosphate-iron complexes. DetergentsPharmaceuticals: as stabilizing and flavoring agentsPersonal Care Products and Cosmetics |
International Chemical Suppliers Archives
September 2016
Categories |









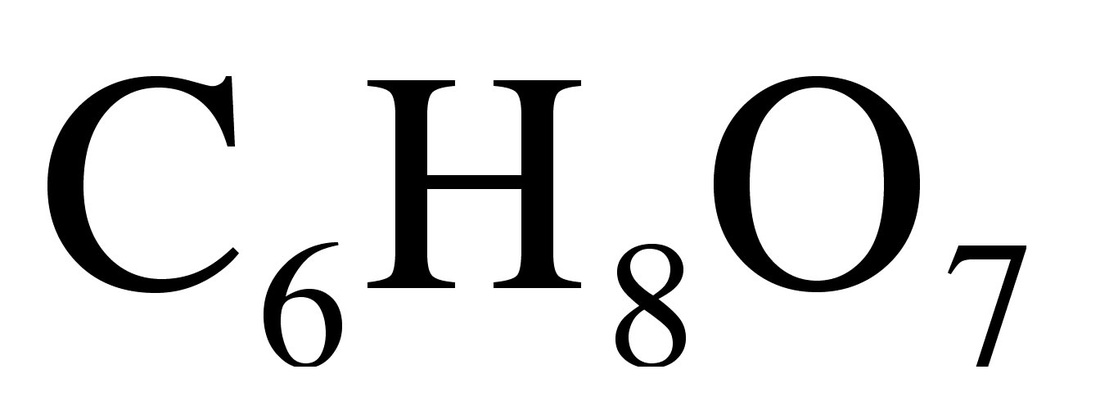


 RSS Feed
RSS Feed
